(888) 4000-234
GO REALISM
Turning Clinical Expertise into Accessible Innovation
We are a hospital namely “ RIDDHIVINAYAK MULTISPECIALITY HOSPITAL (RVMH)” on Plot No 302, Yashwant Gaurav Road, Behind Railway Carshed, Nallasopara (west), Dist. Palghar 401203, Maharashtra State. We are a 200 bedded hospital with 4 lacs sq feet of construction and 5 acres of land, having all the tertiary care services of all major medical specialties. Since the director of the Hospital Dr. Venkat Goyal, is a cardiologist himself, there was an atmosphere of ever increasing interest and pursuit to perfection in Cardiological sciences.
Since the beginning of 21st century there was a growing population of heart failure all over the globe. These were very sick people who required repeated hospitalization. They have a damaged heart and / or lungs who cannot support blood circulation and oxygenation inside the body, so, they required medicines and mechanical support by machines to bailout.
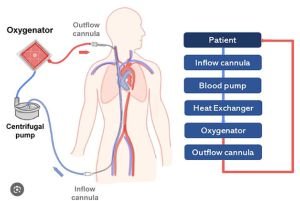
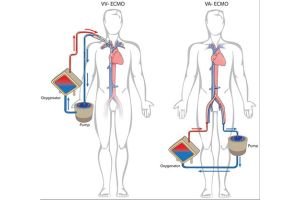
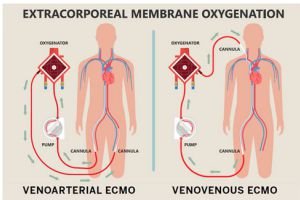
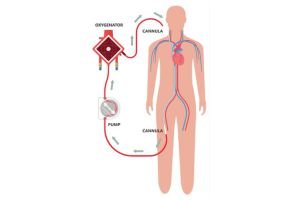
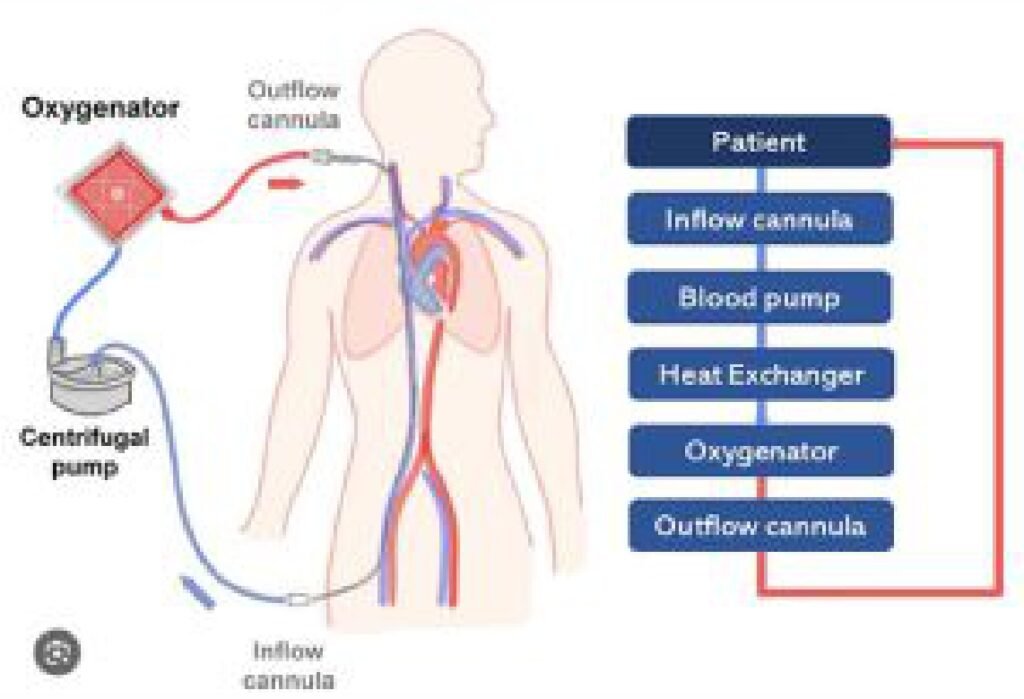
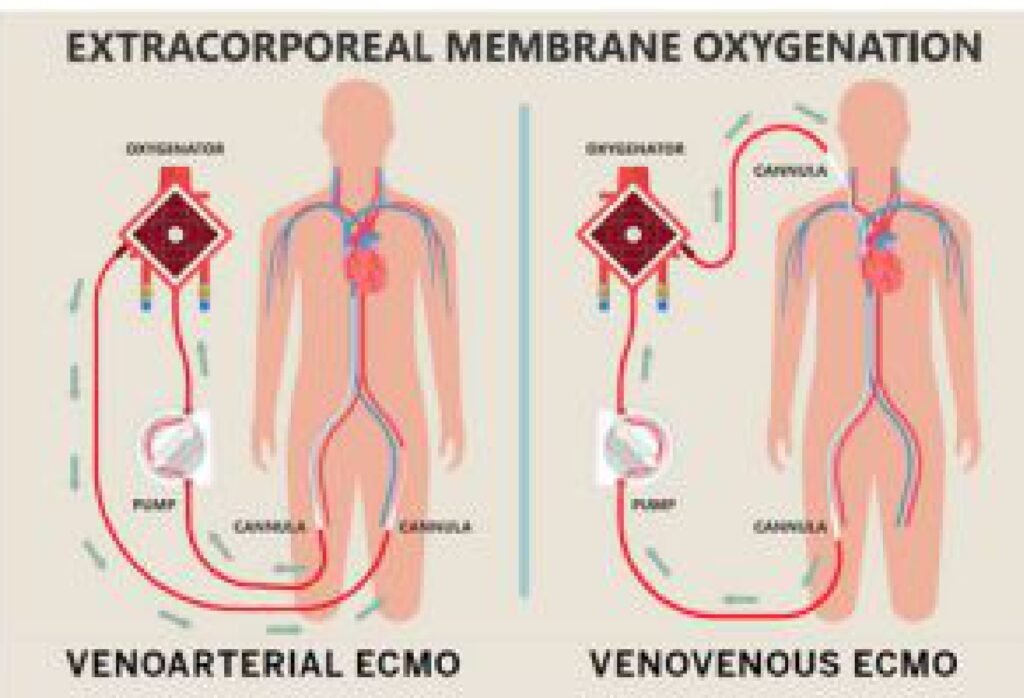
This mechanical support to a failing heart comes under the preview of MCS (Mechanical circulatory support). This machine or in common language “the blood pump” can be intracorporeal (inside the Body) or extracorporeal (outside the body – ECLS : Extracorporeal life support) Under this ECLS science, one of the extracorporeal mechanical support system is called the ECMO (Extra corporeal membrane oxygenation). In common mans understanding this is similar to a Dialysis machine which
The conventional treatment of heart attack i.e blood thinners, coronary angiography and angioplasty or CABG (coronary artery bypass surgery) ends when heart is too weak to revive, and there is where the journey of MCS and organ(heart) transplant starts.
We, at RVMH, having around 25 years of experience in interventional cardiology (angioplasty and bypass surgery) and around 2 decades of experience in MCS. We are pioneers in the country to start ECMO technology since 2006, and start an educational mission of training, promoting and education our fellowmen in ECMO technologies by forming a ECMO Society of India – (ESOI) in 2010.
We are a clinical entity with the use of these equipment for more than 20 years. ECMO science is a very important tool to save lives in case of sudden damage to heart and/ or lungs. But it is very costly (ranging between 15 lacs to 50 lacs of rupees per case) because the entire setup including machines and consumables are imported. So, 9/10 patients needing this support cannot be offered this treatment because of the exuberant cost in Dollar Price.
We are now venturing into indigenous manufacturing this set up in our own country. Over 30 years of clinical practice, now, we have the skills, the experience, the technical details and the wisdom of the positive and negative effects of the device.
Driven by our passion and getting a hope from the Start up Schemes under Atma nirbhar Bharat about the free start up funding and DPIIT Loan schemes we are applying for the financial support of this project.
Challenges We Identified
Imported ECMO and ECLS systems are highly expensive, making advanced care unaffordable for many hospitals.
Limited indigenous R&D infrastructure — from material development to testing and prototyping.
Dependence on foreign technology, restricting customization for Indian clinical needs.
Shortage of skilled personnel and training resources for handling complex imported devices.
Inconsistent service support and delayed spare part availability due to import dependency.
Our Realistic Solutions
Develop cost-effective, Made-in-India ECLS systems that match international quality and reliability.
Build a complete in-house production chain covering design, assembly, and testing for total self-reliance.
Ensure ergonomic, modular, and user-friendly designs tailored for Indian hospital environments.
Establish training and support programs for medical professionals and technicians nationwide.
Maintain strong after-sales and service networks to guarantee uptime and operational efficiency.
Shaping the Future of Life Support Innovation
Our Vision: To pioneer the development of next-generation mechanical support systems for vital organs like the heart, lungs, kidneys, and liver—crafted with precision, built in India, and aligned with global standards. We aim to make Extracorporeal Life Support (ECLS) technology safer, smarter, and accessible to every healthcare institution, ensuring that advanced medical innovation benefits every patient in need.
Our Mission: To serve humanity through innovation and self-reliance, creating affordable and high-quality ECLS systems designed for Indian hospitals. Our mission is to transform clinical expertise into practical, life-saving solutions, bridging the gap between technology and affordability while strengthening India’s position in the global healthcare landscape.
Unique selling points
Economy: The necessity of this ECMO circuit is that the existing imported goods are very costly and the availability of the products is also erratic. Thus, more than 90 % of our population and similar serious patients cannot afford this costly treatment. For every life is precious, and due to lack of resources, they should not be allowed to succumb. SO, if we have an Indian product the cost will definitely be ¼ of the existing market goods
Combination of two components into one will improve ergonomics and cost.
Innovation: New ideas to modify design is a possibility as we have resources to put thoughts into action.
Detail technology overview (given in the powerpoint presentation)
Any Patents/IP you have – NO, BUT WILL BE APPLYING SHORTLY AFTER WE VALIDATE THE CANNULA
Go to market strategy
We are into education, promotion and training of ECMO science on a nonprofitable basis since 2010 under the auspices of ECMO SOCIETY OF INDIA. We have trained more than 1000 highly skilled professionals from nearly 70 corporate hospitals across 20 states in the country. We run the ECMO fellowship courses and certification programs for doctors and paramedics. We have our Journal as Indian Journal of ECMO (IJE) and we are shortly planning to start an Indian College of ECMO. Thus, we have a word in the field to ECMO in India and people do listen to us.
Highlight the uniqueness of your strategy when compared to your competitors: We have a student teacher relationship for most of the ECMO doers in the country. And we have already announced about our efforts for making ECMO circuit in India, so people are eagerly waiting for us to come up with a made in India product which is economical, easily available and had added features apart being multifunctional.
Revenue model
Financial Income source
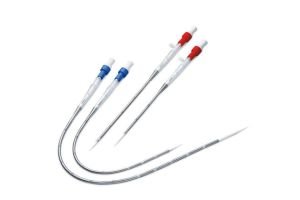
Sale of cannula
Channels of revenue
Self Funding
Bank Loans
Start up Schemes & SEED Funding
Pricing slabs for different customer segments
Export to Asia & Africa
Cost-Effectiveness Analysis
Comparison of pricing with that of competitors absolutely needed
Compare production and selling costs with imported cannula.
Highlight cost savings for hospitals using local cannula (~30-40% cheaper).
Projected Revenue overhead expenses in 3 years
Revenue Diversification
Cannula Sales: 80% of revenue.
After-Sales Support and Training: 10%.
Exports: 10% (from Year 2 onward).
Target market
Data points from credible sources
ELSO data (USA) – 2024 May – No of ECMO done in SW Asia – 5047
ESOI data (India) – 2023 – No of ECMO cases in India 4000 from 200 center
Target Market
All major Hospitals & Medical colleges in India ((583) and 64 PG center))
More than 3,00,000 ICU beds In India
Organ transplant center and CVTS set ups
Asia, Africa and middle east countries
1. Hospital Segments:
Tier-1 Hospitals: Corporate chains like Apollo, Fortis, Medanta, and Max (60% market share).
Tier-2/Tier-3 Hospitals: Regional hospitals expanding critical care facilities, including neonatal and paediatric ECMO.
2. Specialized Centre:
ECMO service providers, heart and lung transplant centre, and paediatric ICUs.
3. Public Healthcare Sector:
Government hospitals under schemes like Ayushman Bharat, promoting affordable ECMO access.
4. Emerging Markets:
Smaller ICUs in tier-2 and tier-3 cities seeking affordable consumables.
5. Exports:
South Asian markets (Bangladesh, Nepal, Sri Lanka), where ECMO use is growing but remains underserved.
Your ideal customer
ECMO center = 200 at present
ECPR (ecmo CPR) population – 75000 at present
Our niche
Critical care Intensivists
Unit economy
Investment to get a customer
- Advertisement
- Demonstration of the technique and training
- Perks – sponsoring a trip to our training program
- Alternative incentives
- Scheme to give alternative consumables as free
- Distribution of medical literature and Books
Customer to purchase of cannula ratio
Individual capacity – 50 cannula / year (5000 intensivists)
- Publicity index – 1: 5 because of a closed loop community and repeated interactions
Business Expansion
- With Indian per capita increase in GDP we feel the deprived group (90 %) which is atleast 45000 cases a year, will be next potential customers. Because ECMO is necessity, not a luxury in critically ill patients
Traction
Transaction Analysis of ECMO Cannula in a Start-Up
Empowering affordable and accessible ECMO therapy in India.
High costs, import dependency, and limited accessibility hinder ECMO adoption in India.
Market Opportunity in India: – The ₹500-700 crore ECMO consumables market is growing at 12-15% annually.
Scope of ECMO in India: Expanding use in cardiac, pulmonary, neonatal, and transplant care, with untapped tier-2/3 potential.
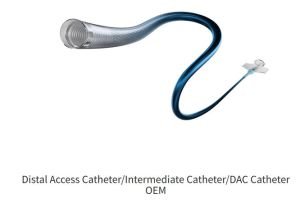
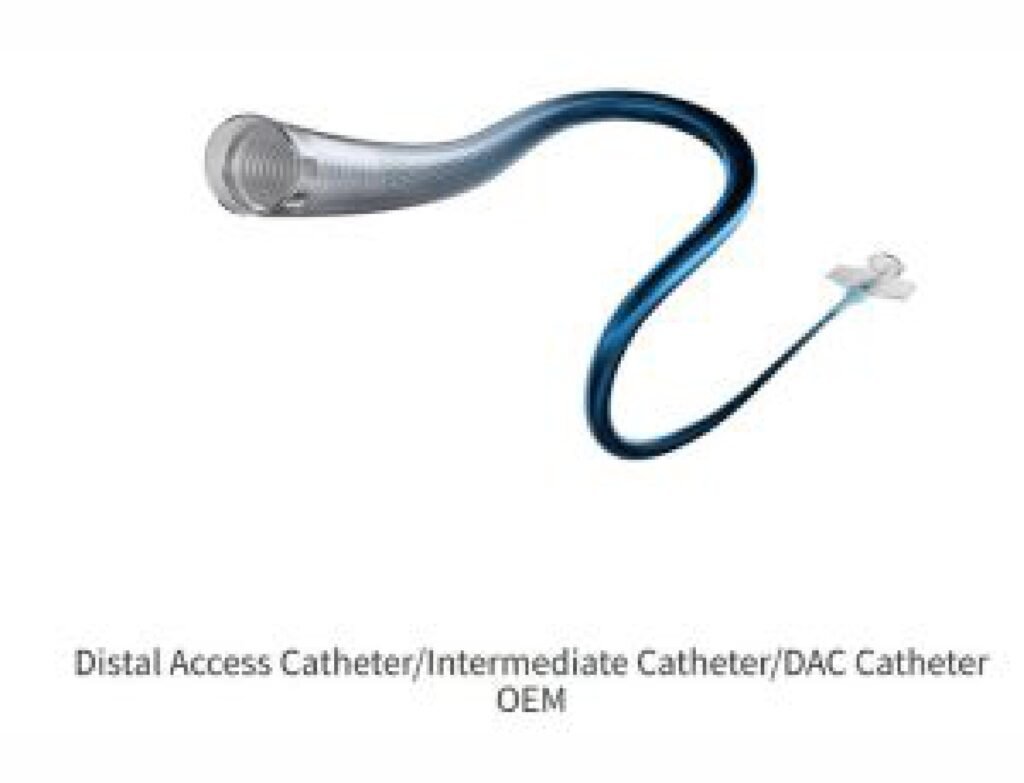
Product Overview: Innovative, biocompatible, and cost-effective ECMO cannulae designed for Indian needs.
Transaction Analysis: Revenue model: B2B sales, government tenders, and collaborations with ECMO providers.
Competitive Landscape (India): 30-40% cheaper than imports, with localized manufacturing for faster delivery.
Go-To-Market Strategy (India): Pilot in metros, scale to tier-2 cities, and secure government healthcare tenders.
Risks & Mitigation: Proactive regulatory planning, strong supplier contracts, and targeted hospital partnerships.
Team: Experienced founders, technical experts, and ECMO-specialist advisors.
Affordable ECMO for Every ICU in India—join us in transforming critical care.

Product or Services
Product Overview
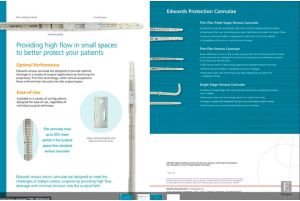
Our ECMO Cannula Features:
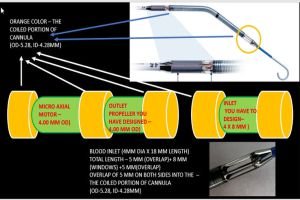
Advanced Flow Dynamics: Ensures higher oxygenation efficiency.
Biocompatibility: Reduces thrombosis, ensuring patient safety.
Cost-Effective Manufacturing: Leveraging local supply chains to minimize costs by 30%.
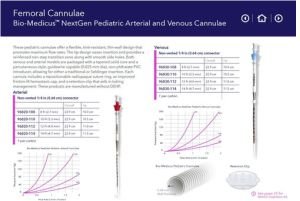
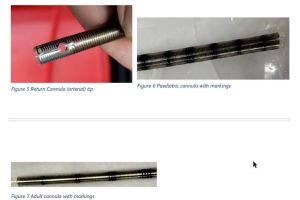
Tailored Designs: Paediatric, neonatal, and adult variants for diverse Indian demographics.
Competitive Landscape (India)
Direct Competitors:
Imported Brands: Expensive and inaccessible in tier-2 and tier-3 markets.
Local Players: Few exist, limited to lower-end consumables.
Our Advantage:
30-40% cost reduction compared to imported cannula.
Custom designs for Indian patient demographics (smaller Paediatric variants).
Reliable local manufacturing, avoiding import delays and forex risks.
Proof of concept
Objective: To validate the design, performance, and market viability of locally manufactured, cost-effective ECMO cannula tailored for the Indian healthcare market.
Key Steps in PoC:
1. Research & Design:
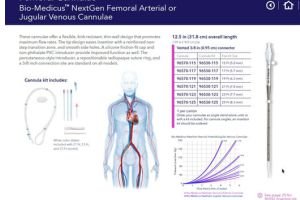
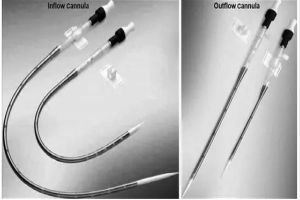
Develop prototypes of venous and arterial ECMO cannula using biocompatible materials.
Focus on flow efficiency, thrombosis prevention, and patient comfort (e.g., pediatric and adult designs).
2. Bench Testing:
Conduct simulations to evaluate flow dynamics, durability, and material compatibility.
Test against international standards (ISO 7198 for vascular prostheses and biocompatibility).
3. Pilot Manufacturing:
Partner with a local medical device manufacturer for small-batch production.
Ensure compliance with Indian regulatory standards (CDSCO) and obtain necessary certifications.
4. Clinical Validation:
Measure clinical outcomes, ease of insertion, and post-procedural complications.
5. Cost-Effectiveness Analysis:
Compare production and selling costs with imported cannula.
Highlight cost savings for hospitals using local cannula (~30-40% cheaper).
6. Stakeholder Feedback:
Gather input from intensivists, ECMO specialists, and perfusionists for design optimization.
Address pain points like delivery time, inventory management, and affordability.
7. Market Validation:
Present results to hospital procurement teams and government healthcare bodies.
Secure initial orders and demonstrate potential demand through MOUs with distributors.
Deliverables:
Functional prototypes that meet clinical and regulatory benchmarks.
Positive feedback and measurable clinical outcomes from pilot trials.
Cost-benefit analysis showing significant savings over imported alternatives.
Stakeholder endorsements and initial purchase commitments.
Timeline:
0-3 Months: Research, design, and prototype development.
4-6 Months: Bench testing and pilot manufacturing.
7-9 Months: Clinical validation and stakeholder engagement.
10-12 Months: Final report, certifications, and market launch.
This POC demonstrates feasibility, clinical reliability, and commercial viability, paving the way for scaling production and market penetration.
Website /Mobile application link and Social Media Link
ESOI (ECMO Society of India)
ECLS Bharat (Extracorporeal Life Support Bharat Pvt. Ltd.)
RVMH (Riddhivinayak Multispeciality Hospital)
Innovative R&D
Affordable Excellence
User-Centric Design
Strong Clinical Foundation
Made in India Initiative
Comprehensive Support
GO REALISM – Key Advantages